Boyle's Law Equation
The mathematical equation for Boyle's Law is:
center;">\[ P \cdot V = k \]
Where:
Explanation of Boyle's Law
Boyle's Law states that, at constant temperature, the pressure of a given mass of gas is inversely proportional to its volume. In simpler terms, when the volume of a gas decreases, the pressure of the gas increases, and vice versa.
Study Guide for Boyle's Law
- Understanding the Equation: Make sure to understand the equation \( P \cdot V = k \) and how pressure and volume are related.
- Graphical Representation: Practice plotting pressure-volume graphs to visualize the inverse relationship described by Boyle's Law.
- Real-life Examples: Look for examples in everyday life where Boyle's Law is evident, such as inflating a balloon or using a syringe.
- Practical Experiments: Perform experiments to demonstrate Boyle's Law using different gases and varying pressure and volume conditions.
- Problem-Solving: Practice solving numerical problems related to Boyle's Law to reinforce understanding of the concept.
By mastering Boyle's Law, you will gain a deeper understanding of the behavior of gases and their properties. These principles are not only important in the field of science, but also have practical applications in various industries.
[Boyle's Law] Related Worksheets and Study Guides:
.◂Science Worksheets and Study Guides Fourth Grade. Cells- The building blocks of living things
Study Guide Cells- The building blocks of living things
Cells- The building blocks of living things  Activity Lesson
Activity Lesson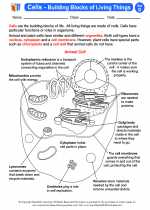 Cells - Building Blocks of Living Things
Cells - Building Blocks of Living Things  Worksheet/Answer key
Worksheet/Answer key Cells- The building blocks of living things
Cells- The building blocks of living things  Worksheet/Answer key
Worksheet/Answer key Cells- The building blocks of living things
Cells- The building blocks of living things  Worksheet/Answer key
Worksheet/Answer key Cells- The building blocks of living things
Cells- The building blocks of living things  Worksheet/Answer key
Worksheet/Answer key Cells - The Building Blocks of Living Things
Cells - The Building Blocks of Living Things  Worksheet/Answer key
Worksheet/Answer key Focus In
Focus In  Vocabulary/Answer key
Vocabulary/Answer key Cells- The building blocks of living things
Cells- The building blocks of living things  Vocabulary/Answer key
Vocabulary/Answer key Cells- The building blocks of living things
Cells- The building blocks of living things  Vocabulary/Answer key
Vocabulary/Answer key Cells- The building blocks of living things
Cells- The building blocks of living things 

 Activity Lesson
Activity Lesson
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Vocabulary/Answer key
Vocabulary/Answer key
 Vocabulary/Answer key
Vocabulary/Answer key
 Vocabulary/Answer key
Vocabulary/Answer key

The resources above cover the following skills:
The Living Environment: Students understand that cells are the basic unit of life, that all life as we know it has evolved through genetic transfer and natural selection to create a great diversity of organisms, and that these organisms create interdependent webs through which matter and energy flow. Students understand similarities and differences between humans and other organisms and the interconnections of these interdependent webs.
Cells: Students describe how living things are made up of one or more cells and the ways cells help organisms meet their basic needs.
Give examples of organisms that consist of a single cell and organisms that are made of a collection of cells.