Charles's Law
Charles's Law is a gas law that describes how gases tend to expand when heated. It states that the volume of a given amount of gas is directly proportional to its absolute temperature, provided that the pressure remains constant.
Key Concepts:
- Volume and Temperature Relationship: According to Charles's Law, as the temperature of a gas increases, its volume also increases, and vice versa, as long as the pressure remains constant.
- Absolute Temperature: Charles's Law uses the absolute temperature scale, typically measured in Kelvin (K), to ensure accurate calculations. The relationship between Celsius (°C) and Kelvin (K) is given by the formula: K = °C + 273.15
- Mathematical Representation: Charles's Law can be expressed as V1 / T1 = V2 / T2, where V1 and T1 are the initial volume and temperature, and V2 and T2 are the final volume and temperature.
Study Guide:
Here are some key points to remember when studying Charles's Law:
- Understand the concept of temperature and its effect on gas volume. Be able to explain how an increase in temperature affects the volume of a gas, and how a decrease in temperature affects the volume.
- Practice converting temperatures between Celsius and Kelvin. Remember that 0°C is equivalent to 273.15 K.
- Be familiar with the mathematical representation of Charles's Law and be able to solve problems using the formula V1 / T1 = V2 / T2.
- Recognize the importance of keeping pressure constant when applying Charles's Law.
By understanding and practicing these key concepts, you will be well-prepared to apply Charles's Law to various gas-related problems and calculations.
.◂Science Worksheets and Study Guides Fifth Grade. Flowers and seeds
Lesson Plan Flowers and Seeds - Bundle
Flowers and Seeds - Bundle  Study Guide
Study Guide Flowers and seeds
Flowers and seeds  Activity Lesson
Activity Lesson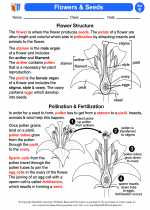 Flowers & Seeds
Flowers & Seeds  Worksheet/Answer key
Worksheet/Answer key Flowers and seeds
Flowers and seeds  Worksheet/Answer key
Worksheet/Answer key Flowers and seeds
Flowers and seeds  Worksheet/Answer key
Worksheet/Answer key Flowers and seeds
Flowers and seeds  Worksheet/Answer key
Worksheet/Answer key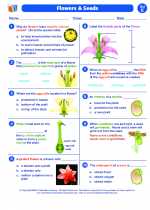 Flowers and seeds
Flowers and seeds  Vocabulary/Answer key
Vocabulary/Answer key Flowers and seeds
Flowers and seeds  Vocabulary/Answer key
Vocabulary/Answer key Flowers and seeds
Flowers and seeds 

 Study Guide
Study Guide
 Activity Lesson
Activity Lesson
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Vocabulary/Answer key
Vocabulary/Answer key
 Vocabulary/Answer key
Vocabulary/Answer key

The resources above cover the following skills:
Life Science
All organisms have structures and systems with separate functions. Students can:
Develop and communicate an evidence-based scientific explanation of the role of different organs or structures that are important for an organism's survival - in both plants and animals