Catalysts
A catalyst is a substance that speeds up a chemical reaction without being consumed in the reaction itself. It works by providing an alternative pathway with a lower activation energy, allowing the reaction to occur more easily and quickly.
Types of Catalysts
There are two main types of catalysts:
- Heterogeneous Catalysts: These are catalysts that are in a different phase from the reactants. For example, a solid catalyst used in a liquid or gaseous reaction.
- Homogeneous Catalysts: These are catalysts that are in the same phase as the reactants. For example, a catalyst dissolved in a liquid reactant.
Role of Catalysts
Catalysts play a crucial role in various industrial processes, such as the production of chemicals, fuels, and pharmaceuticals. They are also essential in biological systems, where enzymes act as catalysts for biochemical reactions in living organisms.
Examples of Catalysts
Some common examples of catalysts include:
- Platinum and palladium used in catalytic converters to reduce harmful emissions from vehicles
- Enzymes in the human body that facilitate digestion and other metabolic processes
- Iron catalysts used in the Haber process to produce ammonia
Importance of Catalysts
The use of catalysts is important because they can significantly increase the rate of a reaction, leading to higher efficiency and lower energy consumption. This has environmental and economic benefits, making processes more sustainable and cost-effective.
Study Tips
To better understand catalysts, consider the following study tips:
- Learn the mechanisms by which catalysts lower activation energy
- Understand the differences between heterogeneous and homogeneous catalysts
- Research specific industrial and biological applications of catalysts
- Practice identifying catalysts in chemical reactions
◂Science Worksheets and Study Guides Fifth Grade. Flowers and seeds

 Study Guide
Study Guide
 Activity Lesson
Activity Lesson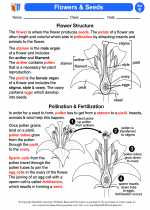
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Worksheet/Answer key
Worksheet/Answer key
 Vocabulary/Answer key
Vocabulary/Answer key
 Vocabulary/Answer key
Vocabulary/Answer key
